also defines: anticlinal, antiperiplanar, clinal, periplanar, synclinal, synperiplanar
https://doi.org/10.1351/goldbook.T06406
In a chain of atoms A-B-C-D, the dihedral angle between the plane containing the atoms A,B,C and that containing B,C,D. In a Newman projection the torsion angle is the angle (having an absolute value between 0° and 180°) between bonds to two specified (fiducial) groups, one from the atom nearer (proximal) to the observer and the other from the further (distal) atom. The torsion angle between groups A and D is then considered to be positive if the bond A-B is rotated in a clockwise direction through less than 180° in order that it may eclipse the bond C-D: a negative torsion angle requires rotation in the opposite sense. Stereochemical arrangements corresponding to torsion angles between 0° and ±90° are called syn (s), those corresponding to torsion angles between ±90° and 180° anti (a). Similarly, arrangements corresponding to torsion angles between 30° and 150° or between -30° and -150° are called clinal (c) and those between 0° and 30° or 150° and 180° are called periplanar (p). The two types of terms can be combined so as to define four ranges of torsion angle; 0° to 30° synperiplanar (sp); 30° to 90° and -30° to -90° synclinal (sc); 90° to 150°, and -90° to -150° anticlinal (ac); ±150° to 180° antiperiplanar (ap). 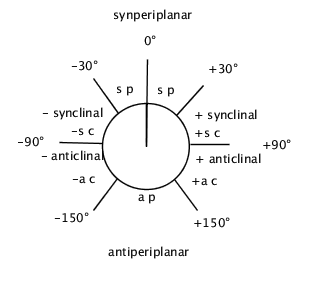 The synperiplanar conformation is also known as the syn- or cis-conformation; antiperiplanar as anti or trans and synclinal as gauche or skew. For macromolecular usage the symbols \(\rm{T}\), \(\rm{C}\), \(\rm{G}^{+}\), \(\rm{G}^{-}\), \(\rm{A}^{+}\) and \(\rm{A}^{-}\) are recommended (ap, sp, +sc, -sc, +ac and -ac respectively).
The synperiplanar conformation is also known as the syn- or cis-conformation; antiperiplanar as anti or trans and synclinal as gauche or skew. For macromolecular usage the symbols \(\rm{T}\), \(\rm{C}\), \(\rm{G}^{+}\), \(\rm{G}^{-}\), \(\rm{A}^{+}\) and \(\rm{A}^{-}\) are recommended (ap, sp, +sc, -sc, +ac and -ac respectively).
