https://doi.org/10.1351/goldbook.A00129
Acyl intermediates include acyl anions, acyl radicals and acyl cations (synonym acylium ions) which are formally derived from oxoacids $\ce{R_{k}E(=O)_{l}(OH)_{m}}$ ($l \neq 0$) by removal of a hydroxyl cation $\ce{HO^{+}}$, a hydroxyl radical $\ce{HO^{.}}$ or a hydroxyl anion $\ce{HO^{-}}$, respectively, and replacement analogues of such intermediates. Acyl anions, radicals and cations can formally be represented by canonical forms having a negative charge, an unpaired electron or a positive charge on the acid-generating element of the oxoacid.
Acyl anions, e.g.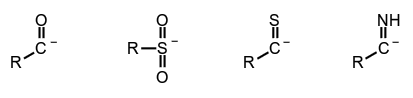
Acyl radicals, e.g.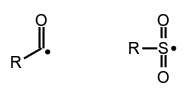
Acyl cations, e.g.
Acyl anions, e.g.
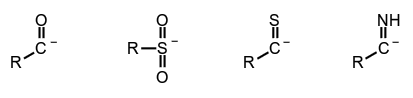
Acyl radicals, e.g.

Acyl cations, e.g.

See also: acyl groups
Sources:
PAC, 1993, 65, 1357. (Revised nomenclature for radicals, ions, radical ions and related species (IUPAC Recommendations 1993)) on page 1357 [Terms] [Paper]
PAC, 1995, 67, 1307. (Glossary of class names of organic compounds and reactivity intermediates based on structure (IUPAC Recommendations 1995)) on page 1312 [Terms] [Paper]
PAC, 1993, 65, 1357. (Revised nomenclature for radicals, ions, radical ions and related species (IUPAC Recommendations 1993)) on page 1357 [Terms] [Paper]
PAC, 1995, 67, 1307. (Glossary of class names of organic compounds and reactivity intermediates based on structure (IUPAC Recommendations 1995)) on page 1312 [Terms] [Paper]