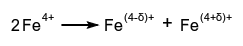https://doi.org/10.1351/goldbook.D01799
- Any chemical reaction of the type , where A, A' and A'' are different chemical species. For example:
 The reverse of disproportionation is called comproportionation. A special case of disproportionation (or 'dismutation') is 'radical disproportionation', exemplified by:
The reverse of disproportionation is called comproportionation. A special case of disproportionation (or 'dismutation') is 'radical disproportionation', exemplified by: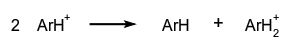 Reactions of the more general type:
Reactions of the more general type: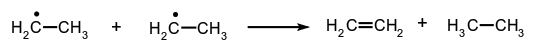 are also loosely described as 'radical disproportionations'.
are also loosely described as 'radical disproportionations'.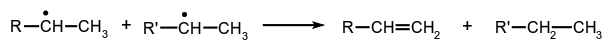
- A reversible or irreversible transition in which species with the same oxidation state combine to yield one of higher oxidation state and one of lower oxidation state. Example: The term also applies to an internal oxidation-reduction process as occurs, for example, among the iron atoms of $\ce{CaFeO3}$, where
 , at Fe subarrays on lowering the temperature.
, at Fe subarrays on lowering the temperature.