https://doi.org/10.1351/goldbook.C01210
A reaction that involves more than one elementary reaction is said to occur by a composite mechanism. The terms complex mechanism, indirect mechanism, and step-wise mechanism are also commonly used. There are two main kinds of evidence for a composite mechanism:
- The kinetic equation for the reaction does not correspond to its stoichiometry.
- There is experimental evidence, direct or indirect, for intermediates of such a nature that it is necessary to conclude that more than one elementary reaction is involved.
- Reactions occurring in parallel, such as:
 are called parallel reactions or simultaneous reactions. When there are simultaneous reactions there is sometimes competition, as in the scheme:
are called parallel reactions or simultaneous reactions. When there are simultaneous reactions there is sometimes competition, as in the scheme:
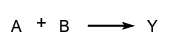 where B and C compete with one another for A.
where B and C compete with one another for A.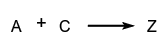
- Reactions occurring in forward and reverse directions are called opposing reactions:
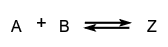
- Reactions occurring in sequence, such as are known as consecutive reactions.

- Reactions are said to exhibit feedback if a substance formed in one step affects the rate of a previous step. For example, in the scheme: The intermediate Y may catalyse the reaction
 (positive feedback) or it may inhibit it (negative feedback).
(positive feedback) or it may inhibit it (negative feedback).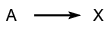
- Chain reactions