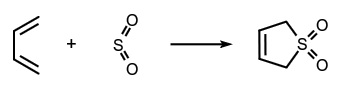https://doi.org/10.1351/goldbook.C01014
A form of cycloaddition across the terminal atoms of a fully conjugated system with formation of two new $\ce{\upsigma\!\mbox{-}bonds}$ to a single atom of the ('monocentric') reagent. There is formal loss of one $\ce{\uppi\!\mbox{-}bond}$ in the substrate and an increase in coordination number of the relevant atom of the reagent. An example is the addition of sulfur dioxide to butadiene: 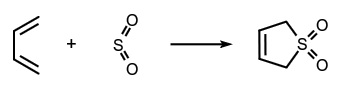 The reverse of this type of reaction is designated 'cheletropic elimination'.
The reverse of this type of reaction is designated 'cheletropic elimination'.