https://doi.org/10.1351/goldbook.C00964
If carbon atoms 1, 2, 4 and 5 of cyclohexane occupy coplanar positions and when carbon atoms 3 and 6 are on opposite sides of the plane the conformation (of symmetry group $D_{\rm{3d}}$) is called a chair form. The same term is applied to similar conformations of analogous saturated six-membered ring structures containing hetero-atoms and/or bearing substituent groups, but these conformations may be distorted from the exact $D_{\rm{3d}}$ symmetry. For cyclohexane and most such analogues, the chair form is the most stable conformation. If the cyclohexane conformation has no centre of symmetry but possesses two planes of symmetry, one of them bisecting the bonds between atoms 1 and 2 and between 4 and 5 and the other plane passing through atoms 3 and 6 (which lie out of the plane and on the same side of the plane containing 1, 2, 4 and 5), that conformation (of symmetry group $C_{\rm{2v}}$) is called a boat form and it is generally not a stable form. Again, this term is also applied to structural analogues. The conformation of $D_{2}$ symmetry passed through in the interconversion of two boat forms of cyclohexane is called the twist form (also known as skew boat, skew form and stretched form). 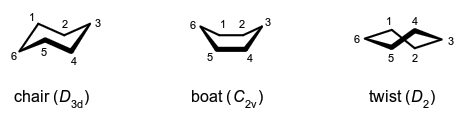 In a five-membered ring a conformation in which two adjacent atoms are maximally displaced, in opposite directions, relative to the plane containing the other three carbon atoms has been called a half-chair but is better called a twist conformation.
In a five-membered ring a conformation in which two adjacent atoms are maximally displaced, in opposite directions, relative to the plane containing the other three carbon atoms has been called a half-chair but is better called a twist conformation.
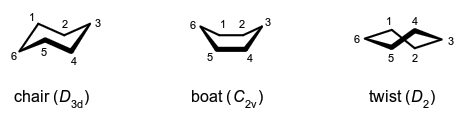
Note: In carbohydrate chemistry the term twist refers to a five-membered ring and the $D_{2}$ symmetry six-membered ring is referred to as skew.
See also: envelope conformation, half-chair