https://doi.org/10.1351/goldbook.A00266
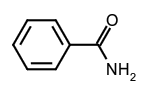
- Derivatives of oxoacids $\ce{R_{k}E(=O)_{l}(OH)_{m}}\ (l \neq 0)$ in which an acidic hydroxy group has been replaced by an amino or substituted amino group. Chalcogen replacement analogues are called thio-, seleno- and telluro-amides. Compounds having one, two or three acyl groups on a given nitrogen are generically included and may be designated as primary, secondary and tertiary amides, respectively, e.g. benzamide,
 N,N-dimethylmethanesulfonamide,
N,N-dimethylmethanesulfonamide,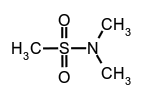 secondary amides (see imides),
secondary amides (see imides),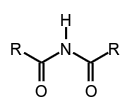 tertiary amides,
tertiary amides,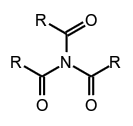 phenylphosphonamidic acid.
phenylphosphonamidic acid.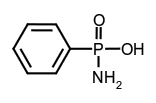
Notes:- Amides with $\ce{NH2}$, $\ce{NHR}$ and $\ce{NR2}$ groups should not be distinguished by means of the terms primary, secondary and tertiary.
- Derivatives of certain acidic compounds $\ce{R_{n}E(OH)_{m}}$, where $\ce{E}$ is not carbon (e.g. sulfenic acids, $\ce{RSOH}$, phosphinous acids, $\ce{R2POH)}$, having the structure $\ce{R_{n}E(NR2)_{m}}$ may be named as amides but do not belong to the class amides proper, e.g. $\ce{CH3CH2SNH2}$ ethanesulfenamide or ethylsulfanylamine.
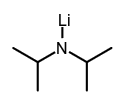
- The term applies also to metal derivatives of ammonia and amines, in which a cation replaces a hydrogen atom on nitrogen. Such compounds are also called azanides, e.g. lithium diisopropylamide, synonym lithium diisopropylazanide.
See also: carboxamides, lactams, peptides, phosphoramides, sulfonamides