- The physical processes frequently studied in polymer science are crystallisation, melting, and the glass transition. See the figures below, which illustrate the corresponding characteristic temperatures. The ordinate used in the figures is specific heat capacity \(c_{p}/\pu{J K-1 g-1}\). When using DSC, the quantity measured is the specific heat flow rate/\(\pu{W g-1}\), and when using DTA, the quantity measured is the temperature difference between sample and reference. Both quantities can be converted to \(c_{p}\) by calibration.

Figure 1: Characteristic temperatures found from a DSC or DTA cooling experiment involving crystallisation. In a heating experiment, the subscripts for "initial" and "final" are interchanged. Cold crystallisation can sometimes be observed in a heating experiment. \(T_{\rm{i,cr}}\) = initial (onset) crystallisation temperature (at which crystallisation starts). \(T_{\rm{ei,cr}}\) = extrapolated initial (onset) crystallisation temperature. \(T_{\rm{p,cr}}\) = peak crystallisation temperature (at which the maximum rate of crystallisation occurs). \(T_{\rm{ef,cr}}\) = extrapolated final (end) crystallisation temperature. \(T_{\rm{f,cr}}\) = final (end) crystallisation temperature (at which crystallisation stops). \(\Delta_{\rm{cr}} h\) = specific enthalpy of crystallisation, unit: \(\pu{J g-1}\). \(c_{p}\) = specific heat capacity, unit: \(\pu{J K-1 g-1}\).
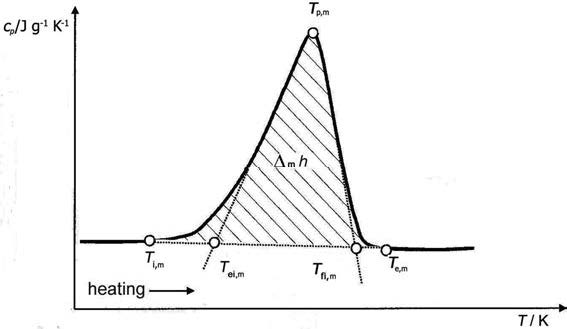
Figure 2: Characteristic temperatures found from a DSC or DTA heating experiment involving melting (fusion). _028101_ = initial (onset) melting temperature (at which melting starts). _028102_ = extrapolated initial (onset) melting temperature. _028103_ = peak melting temperature (at which the maximum rate of melting occurs). _028104_ = extrapolated final (end) melting temperature. _028105_ = final (end) melting temperature (at which melting stops). _028106_ = specific enthalpy of fusion, unit: \(\pu{J g-1}\). \(c_{p}\) = specific heat capacity, unit: \(\pu{J K-1 g-1}\).

Figure 3: Characteristic temperatures found from a DSC or DTA experiment involving a glass transition. The experiment is often carried out by cooling the polymer melt rather than heating the polymer glass because of the small variation of the transition temperature with the cooling rate in contrast to the more complex situation in a heating experiment; see enthalpy relaxation. _028107_ = initial (onset) temperature of the glass transition. _028108_ = extrapolated initial (onset) temperature of the glass transition. _014497_ = mid-point temperature, equated to the glass-transition temperature. _028109_ = extrapolated final (end) temperature of the glass transition. _028110_ = final (end) temperature of the glass transition. \(c_{p}\) = specific heat capacity, unit: \(\pu{J K-1 g-1}\). _028111_ = change in specific heat capacity, unit: \(\pu{J K-1 g-1}\).
- The glass transition is usually near the point of inflection of the function \(c_{p}(T)\).
- In a symmetrical curve, the point of inflection is at \(½\) of the height of the step of the curve (the mid-point temperature).
- Alternative methods for evaluating the glass-transition temperature, \(T_{\rm{g}}\), from DSC or DTA curves exist, for example, evaluation of the temperature of equal specific enthalpy of the glass and the melt.