https://doi.org/10.1351/goldbook.08883
Polymers prepared exclusively from aniline, or substituted aniline, or both in admixture (source-based class name).
Notes:
- Examples of polyanilines (PANI) in the leucoemeraldine base form (see Note 2) are: (the first name is the structure-based while the second one is the source-based name of the polymer).
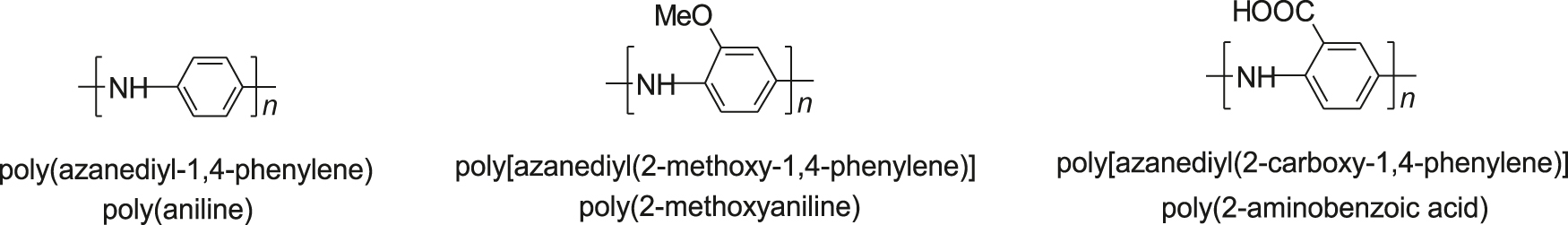
- Polyanilines exist in various forms (see scheme) differing in the degrees of oxidation (p-doping) and protonation (proton doping). Gem-based names (leucoemeraldine, emeraldine, pernigraniline) were given to particular PANI forms in 1910 by Green and Woodhead.
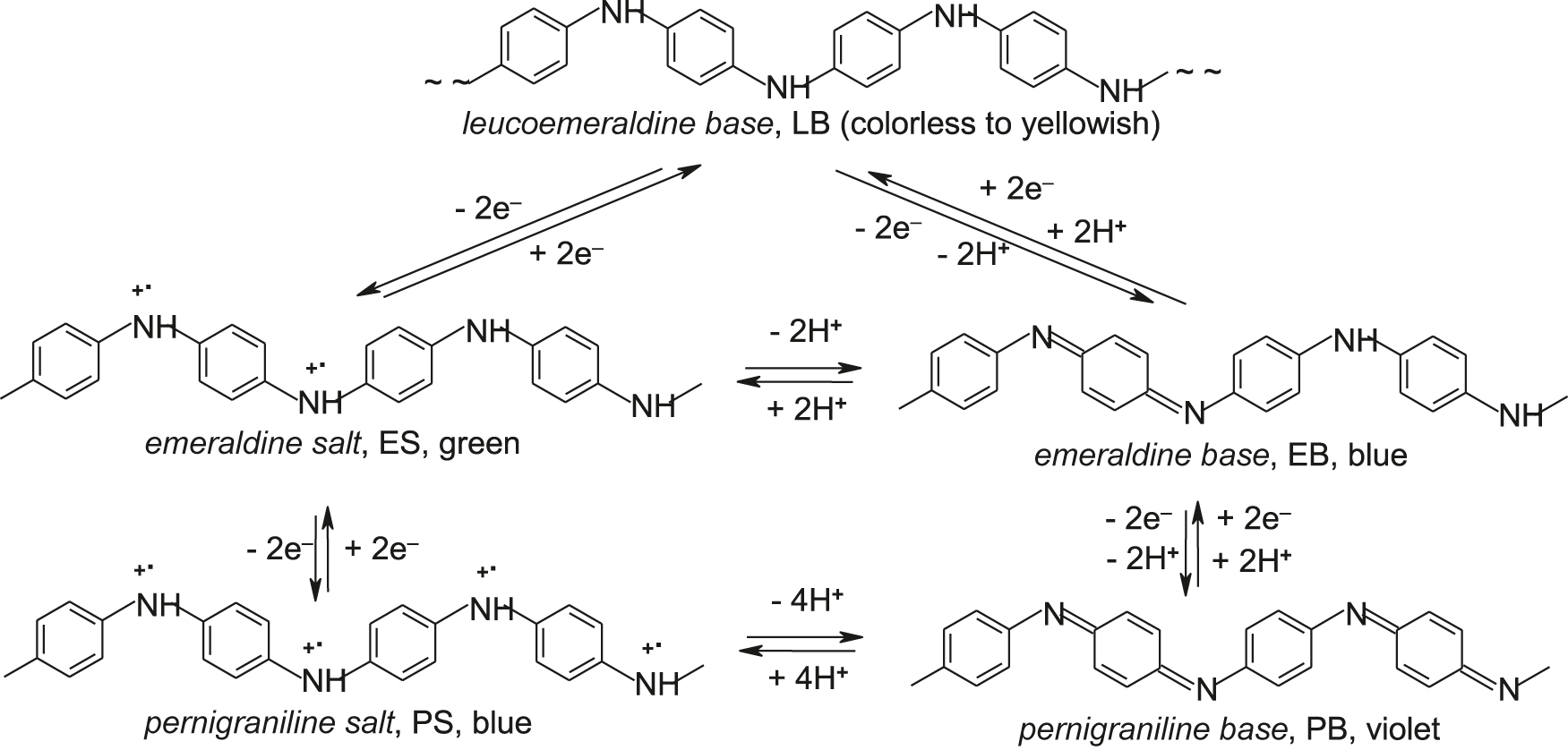
- Redox doping of PANI-LB by the electron transfer (left branch in the scheme) directly gives PANI salts ES and PS, whereas dehydrogenation of LB (right branch in the scheme) gives PANI bases: EB and PB. PANI bases are reversibly transformed to corresponding PANI salts by proton doping. The intermediate oxidation state forms, protoemeraldine (product of one-electron oxidation) and nigraniline (product of three-electron oxidation of LB) are sometimes recognized in the literature.
- PANIs carrying strongly acidic group such as sulfonic or phosphonic group attached to benzene rings are referred to as self-doped PANIs, since the attached acidic groups afford protons ensuring proton doping of main chains.
- The PANI ES form is an intrinsically conducting polymer (conductivity of \(\pu{1-10 S cm-1}\)) in which positive polarons and protons are major charge carriers. It is easy accessible by electrochemical or chemical oxidation of anilines in an acidic aqueous medium and it found many applications in electrochemical and photovoltaic cells, batteries, displays and others.